IV Cannula Gauge Selection Guide: 14G to 24G Clinical Use Cases
Published May 8, 2026 · 12 min read · By HEZE YINUO MEDICAL
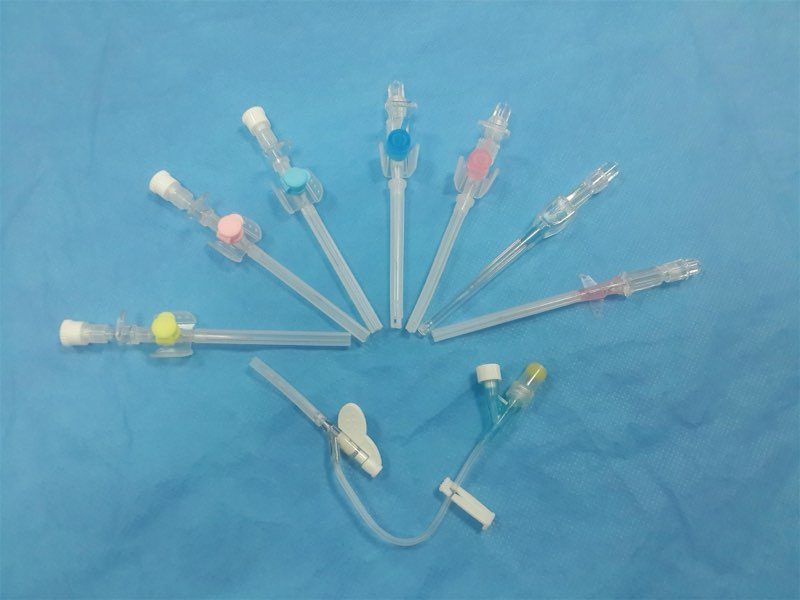
Quick orientation. The peripheral IV cannula is one of the most heavily used disposable medical devices in any hospital — and one of the most procurement-sensitive, because the wrong gauge mix produces clinical compromises (haemolysis on transfusion, vein damage on paediatrics, inadequate flow on resuscitation) that compound across thousands of insertions per ward per month. This guide walks through ISO 6009 colour coding, gauge-by-gauge clinical use cases, flow rates, length options, material choices, safety variants, and the procurement specifications that should drive your IV-cannula SKU mix.
1. Anatomy of an IV cannula
A peripheral IV cannula has six functional components:
- Stainless-steel introducer needle — used to puncture the vein, then withdrawn and discarded. Bevel geometry (back-cut or three-facet) affects insertion force.
- Cannula tube — the soft plastic catheter that remains in the vein. FEP, PUR or PTFE.
- Hub — the colour-coded plastic connector that mates with the IV line. ISO 6009 standardises the colour by gauge.
- Wings — flat plastic flanges to anchor the cannula against the skin during taping.
- Injection port (optional) — a side port with rubber septum allowing direct medication injection into the cannula without disconnecting the IV line.
- Flashback chamber — translucent reservoir at the back of the needle that fills with blood when the vein is successfully entered, confirming placement.
- Safety mechanism (on safety variants) — passive shield or retracting needle that engages after withdrawal, preventing needlestick injury.
Our IV cannulas are produced across the full 14G–26G range under ISO 6009 colour coding, with FEP cannula tubes, with-port and without-port variants, and safety mechanism options.
2. ISO 6009 colour coding
ISO 6009 is the international standard for colour coding of hub fittings on hypodermic and IV needles. Compliant manufacturers use the colours below universally; clinicians can identify the gauge by hub colour at a glance, which matters in emergency settings where reading printed text is too slow.
| Gauge | ISO 6009 colour | Outer diameter (approx.) | Gravity flow (water) |
|---|---|---|---|
| 14G | Orange | 2.10 mm | ~240–300 ml/min |
| 16G | Grey | 1.70 mm | ~180–200 ml/min |
| 18G | Green | 1.30 mm | ~80–100 ml/min |
| 20G | Pink | 1.10 mm | ~55–65 ml/min |
| 22G | Blue | 0.90 mm | ~32–36 ml/min |
| 24G | Yellow | 0.70 mm | ~20–24 ml/min |
| 26G | Purple | 0.60 mm | ~13–15 ml/min |
Flow rates above are approximate gravity-fed water flow at standard cannula length. Actual clinical flow with viscous fluids (blood products, lipid emulsions) is meaningfully lower; rapid-infusion settings use pressure infusion or pumps to achieve target rates.
3. Gauge-by-gauge clinical use cases
14G (orange) — Trauma resuscitation
The largest gauge in routine clinical use. Flow ≥240 ml/min permits massive transfusion in trauma, GI haemorrhage and obstetric haemorrhage cases. 14G is uncomfortable for patients at insertion and difficult to place in normal-calibre veins — it is reserved for resuscitation use cases where flow rate dominates. Stock heavily in trauma centres, OB units, and emergency departments.
16G (grey) — Surgical and major procedures
The default for surgical patients undergoing procedures with anticipated blood loss, major orthopaedic surgery, and ICU patients on high-volume fluid management. Flow ~180 ml/min supports rapid transfusion at lower viscous shear than 18G, reducing haemolysis risk for blood products. Stock in OR, ICU, ED.
18G (green) — Blood transfusion and standard adult IV
The most widely used gauge for adult inpatient IV access. Flow ~80–100 ml/min supports blood transfusion (the minimum gauge generally recommended), contrast injection for CT imaging, and most therapeutic infusions. Comfortable for most patients with reasonable vein calibre. Stock heavily across all adult wards.
20G (pink) — Routine adult IV
The volume-leading gauge for routine adult IV access where blood transfusion is not anticipated. Flow ~60 ml/min is sufficient for most antibiotics, fluids, electrolytes and standard medications. Better patient comfort than 18G. Stock heavily on general medical and surgical wards.
22G (blue) — Paediatric, geriatric, fragile veins
Standard for paediatric patients beyond infancy, elderly patients with fragile or difficult veins, and oncology patients after multiple cannulation cycles. Flow ~36 ml/min is adequate for most non-emergent infusion; not recommended for blood transfusion or rapid fluid resuscitation. Stock heavily in paediatrics, geriatrics, oncology.
24G (yellow) — Neonates, very fragile veins
Used in neonatal, paediatric infants, and adults with extremely fragile veins (eg. severe oncology cachexia). Flow ~22 ml/min restricts use to drug delivery and small-volume infusion; not for transfusion or rapid replacement. Stock in NICU, paediatric units, palliative care.
26G (purple) — Specialty neonatal use
Smaller still, for the smallest neonates or specific specialty applications. Not part of every hospital's stock; specialty supply.
4. Length options
Cannula length should match target vein calibre and depth. Common length offerings:
- 14G: 45 mm (single length common)
- 16G: 32 mm or 45 mm
- 18G: 32 mm or 45 mm
- 20G: 25 mm or 32 mm
- 22G: 25 mm
- 24G: 19 mm or 25 mm
- Specialty: 50 mm 18G for upper-arm "long peripheral" / midline access where extended dwell is anticipated
Using a too-long cannula in a small vein increases through-puncture risk and post-insertion bleeding. Procurement should match length-by-gauge SKUs to the wards using them.
5. Material choices: FEP vs PUR vs PTFE
- FEP (fluorinated ethylene propylene) — the dominant cannula tube material globally. Smooth surface, kink-resistant, biocompatible. Maintains shape at body temperature. Volume-leading material for most adult and paediatric cannulas.
- PUR (polyurethane) — softens at body temperature, conforming to the vein curve. Reduces vein irritation and supports longer dwell times (typically 72–96 hours vs 48–72 for FEP). Used in higher-end product lines, oncology and specialty applications.
- PTFE (polytetrafluoroethylene) — the original cannula material. Excellent biocompatibility but stiffer than FEP. Used in some specialty and legacy designs; less common in current high-volume production.
For most procurement, FEP is the default. PUR is a valuable upgrade for oncology, palliative care and other settings with extended dwell requirements, at a 20–40% unit-cost premium.
6. Safety variants and the cost-benefit calculus
Safety IV cannulas integrate a passive needle-shielding mechanism that automatically engages after withdrawal, preventing needlestick injury. The mechanism varies by manufacturer — passive shield, retracting needle, sliding cover — but all serve the same purpose.
Regulatory drivers:
- US — Needlestick Safety and Prevention Act (2000) requires safety devices in patient care
- EU — Council Directive 2010/32/EU on prevention of sharp injuries in healthcare
- Many other markets adopting similar regulation, particularly in tertiary hospitals
Cost premium: 1.5–3× standard non-safety cannula. Benefit: needlestick injury reduction (occupational health saving) and tender-eligibility for regulated markets.
For US, EU and tier-one Asian hospital procurement, safety cannulas are typically mandatory. For unregulated or developing markets, the cost premium is balanced against occupational health priority. Many tenders now specify safety as the default and require a documented exemption to use non-safety.
7. Bulk procurement specifications
SKU mix recommendations
A typical adult inpatient ward stocks the following gauge mix (approximate share of consumption):
- 20G — 40–50% (volume leader)
- 18G — 25–30%
- 22G — 15–20%
- 16G — 5–8%
- 14G — 1–3%
Paediatric and neonatal wards invert the mix toward 22G and 24G. Trauma and ICU shift toward 16G and 18G. Confirm hospital-specific consumption before placing initial orders, then refine on real-world usage.
Unit cost ranges (FOB China)
- Standard non-safety 14G–24G IV cannula: USD 0.07–0.20 per piece
- With injection port: +USD 0.02–0.05 per piece
- Safety variant: USD 0.15–0.50 per piece (1.5–3× non-safety)
- PUR cannula tube upgrade: +USD 0.03–0.07 per piece
MOQ and lead time
Typical MOQ is 100,000 pieces per gauge per length per production run, sometimes scaling down to 50,000 for less common gauges (14G, 26G). Safety variants typically MOQ 200,000+ due to safety-mechanism module assembly logistics. Lead time 25–35 days production. Standard packaging: individual blister, 50 / 100 per inner box, 1000 per master carton; sterile EO-packed. Shelf life 36 months from manufacturing date.
8. Common procurement pitfalls
- Not specifying ISO 6009 colour coding. Some legacy or regional manufacturers use slightly different colours. Specify ISO 6009 compliance to avoid clinical confusion.
- Mismatched gauge / length combination. Specifying "all 18G at 32 mm" without considering anatomic variation. Stock multiple lengths in the high-volume gauges (18G, 20G, 22G).
- Underestimating safety variant requirement. Tender language often says "safety" but doesn't specify the mechanism type. Different mechanisms have different clinician-acceptance levels — pilot one or two before committing volume.
- Not splitting paediatric and adult inventory. Paediatric ward consuming 22G/24G heavily but ordered against an adult-mix template runs out of small gauges and overstocks 18G/20G.
- Counterfeit colour coding. Some sub-spec product mimics ISO colours but uses inconsistent dye batches that fade in storage. Demand colour-fastness in your QC spec.
- Ignoring catheter material for long-dwell cases. FEP is fine for 48–72 hour dwell; PUR is worth the premium for oncology and palliative care where dwell exceeds 72 hours.
9. Frequently asked questions
What is the standard colour coding for IV cannula gauges?
ISO 6009 standardises the colour coding for hypodermic and IV cannula hubs: 14G is orange, 16G is grey, 17G is white, 18G is green, 20G is pink, 22G is blue, 24G is yellow, 26G is purple. The colour coding is universal across compliant manufacturers and helps clinicians select the correct gauge by sight under emergency conditions. Some legacy or regional manufacturers use slightly different colour conventions; for hospital procurement specify ISO 6009 compliance to avoid mismatch.
Which IV cannula gauge should I use for blood transfusion?
Blood transfusion typically uses 18G (green) or 16G (grey) IV cannulas. 18G is the minimum gauge widely recommended to avoid haemolysis; 16G is preferred for rapid transfusion or in trauma settings. Larger gauges (14G, 16G) are mandatory for massive transfusion protocols. Smaller gauges (20G, 22G) can be used in stable patients with a slow transfusion rate, but increase the risk of haemolysis from shear stress at the cannula tip.
What gauge is appropriate for paediatric and neonatal IV access?
Paediatric IV access typically uses 22G (blue) or 24G (yellow) cannulas; neonatal access uses 24G or smaller. The very small vein calibre and high fragility in young children mean the largest cannula that fits is the wrong choice — clinical guidelines emphasise smallest-effective-gauge for vein preservation. 26G cannulas are available for the smallest neonates but are not part of every hospital's stock. For paediatric units, ensure your inventory is heavily weighted toward 22G and 24G.
What are the common materials used in IV cannulas?
The cannula tube (the part that stays in the vein) is typically made of FEP (fluorinated ethylene propylene), PUR (polyurethane), or PTFE (polytetrafluoroethylene). FEP is the most common — biocompatible, kink-resistant, with smooth surface finish. PUR softens at body temperature, reducing vein irritation and is often used for longer-dwell catheters. PTFE is the original material, still in some specialty designs. The needle (used only for vein insertion, then withdrawn) is stainless steel with siliconised coating to reduce insertion force.
Are safety IV cannulas worth the cost premium?
Safety IV cannulas — with passive needle-shielding mechanisms that automatically retract or cover the needle after insertion — are required by occupational-safety regulation in the US (Needlestick Safety and Prevention Act, 2000) and many EU member states. They cost 1.5–3× more than standard non-safety cannulas at factory gate. For hospitals in regulated markets, safety variants are mandatory; for unregulated settings the cost premium is balanced against needlestick-injury reduction value. Many tenders now specify safety as the default.
What length options are common for IV cannulas?
Standard IV cannula lengths range from 19 mm to 50 mm. The most common are: 14G — 45 mm; 16G — 32 mm or 45 mm; 18G — 32 mm or 45 mm; 20G — 25 mm or 32 mm; 22G — 25 mm; 24G — 19 mm or 25 mm. Length should be matched to the target vein calibre; using too long a cannula in a small vein increases risk of through-puncture. Specialty length options (e.g. 50 mm 18G for upper-arm midline access) exist as separate SKUs.
What is the typical MOQ for IV cannulas from a Chinese manufacturer?
Typical MOQ for non-customised IV cannulas is 100,000 pieces per gauge per length per production run, sometimes scaling down to 50,000 for less common gauges (14G, 26G) or specialty length variants. Safety IV cannulas typically have a higher MOQ (200,000+) because the safety mechanism module assembly line runs in larger batches. For OEM packaging, a one-time tooling fee applies but per-unit MOQ is unchanged. We carry the full 14G–24G range under ISO 6009 colour coding, available with and without injection ports, and in safety variants.
10. Summary and how to request a quote
IV cannula gauge selection is one of the most consequential SKU decisions in any hospital's disposable supply chain. Match gauge to clinical scenario, length to vein calibre, material to dwell time, and safety mechanism to occupational health regulation. The right SKU mix changes by ward — paediatric weight toward 22G/24G, trauma toward 16G/18G, general ward weight toward 18G/20G.
As a manufacturer of peripheral IV cannulas across the full 14G–26G range, plus the broader needles family — hypodermic needles, scalp vein / butterfly sets, blood collection needles, blunt tip / blunt fill needles, safety blood lancets — we ship to hospital networks and distributors in 50+ countries. Send us your gauge mix, length preference, safety / non-safety, port / non-port, target quantity and destination market, and we will respond within one working day with applicable certifications, MOQ, lead time and a tiered quote. Request a quote
