Infusion Set Components, Types & Compatibility: A Complete Guide
Published May 6, 2026 · 15 min read · By HEZE YINUO MEDICAL
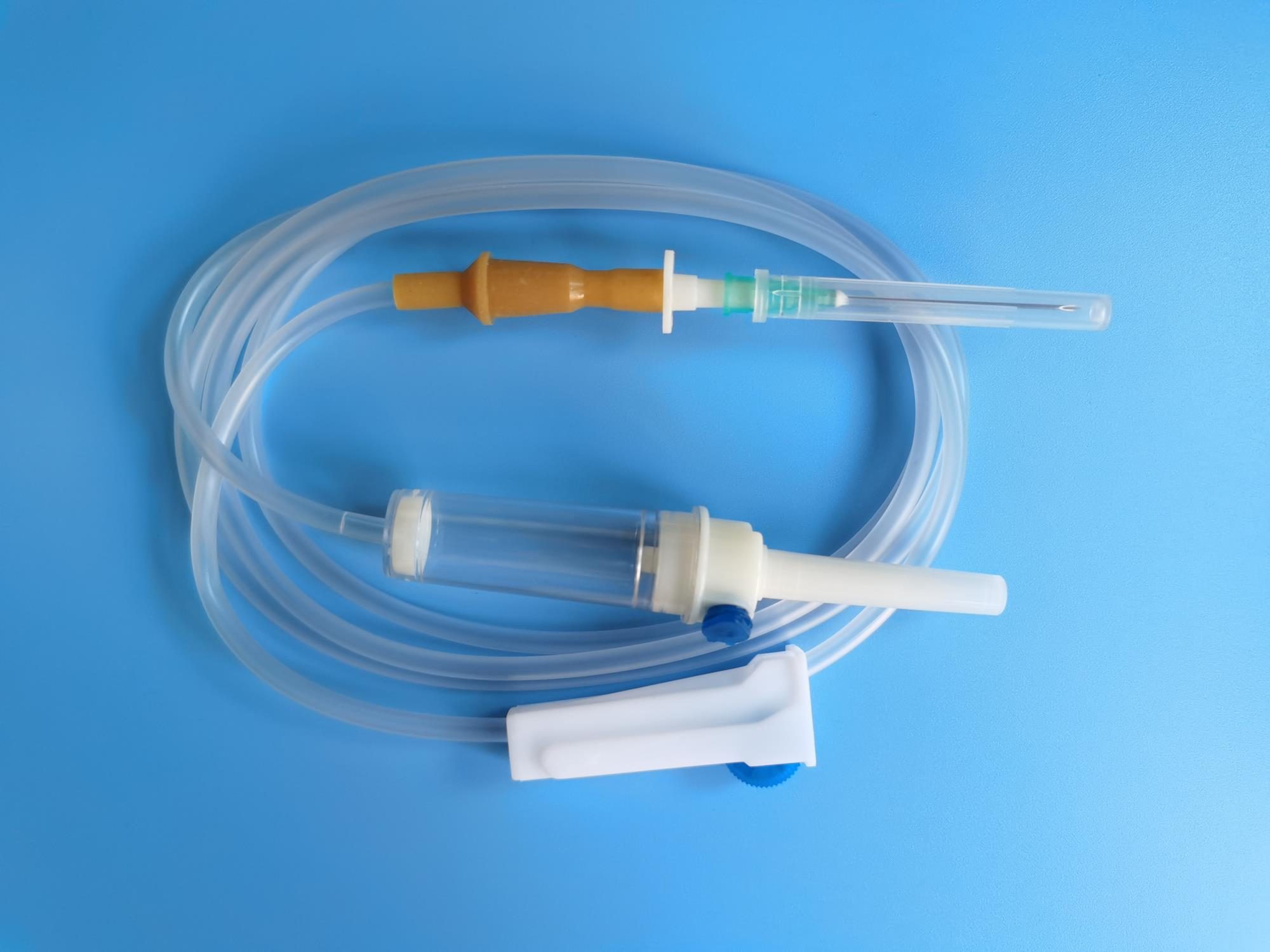
Quick orientation. An IV infusion set is a single-use disposable assembly that delivers fluid from a bag or bottle to a patient under gravity or pump pressure. Across an annual hospital procurement, the catalogue typically covers six to ten distinct set types — each defined by its drop factor, connector, side-port configuration, in-line filter, plasticiser system, and the ISO standard it is tested against. Mixing them up at the spec stage causes audit failures, compatibility errors with IV cannulas and pumps, and replacement orders that destroy your unit-cost gains. This guide walks through the components, the type families, and the compatibility rules so you can write specifications that produce correct quotes the first time.
1. The standard reference: ISO 8536-4 and ISO 1135-4
Two ISO standards govern most of the disposable infusion-set market:
- ISO 8536-4 — "Infusion equipment for medical use, Part 4: Infusion sets for single use, gravity feed". This is the umbrella standard for everyday IV infusion sets used with bags, bottles, and gravity drip stands. It defines geometry, materials, sterility, drip-chamber transparency, drop former dimensions, and labelling.
- ISO 1135-4 — "Transfusion equipment for medical use, Part 4: Transfusion sets for single use, gravity feed". The parallel standard for blood transfusion. The geometry overlaps with 8536-4 but blood sets carry a 170–200 micron screen filter inside the drip chamber and have wider tubing.
Other parts of the ISO 8536 family (Parts 8, 9, 10, 12, 13) cover pump-driven sets, burette sets, sets for use with infusion pumps, and accessory devices. When a tender specifies "ISO 8536-4 compliant", it means the set must pass the gravity-feed performance tests; when it says "ISO 8536-8" or "ISO 8536-10", a different test set applies. Read the standard reference on the spec sheet, not the marketing claim.
2. Anatomy of a standard infusion set
From the IV bag to the patient, a typical gravity-feed infusion set has the following components in order:
Piercing spike (with or without air vent)
A rigid moulded ABS or polypropylene tip that punctures the rubber stopper of an IV bag or the cap of a glass bottle. Vented spikes have a small one-way air filter that lets atmospheric air enter the bottle as fluid drains; non-vented spikes rely on the IV bag collapsing under negative pressure. Match the spike to the container: vented for rigid bottles, non-vented for flexible PVC bags. Most modern sets ship with a removable air-vent cap so the same set works on either container.
Drip chamber
A transparent flexible chamber, normally 50–80 mm tall, that lets the clinician visually confirm flow and trap air bubbles before they reach the patient. The chamber sits below the spike and above the tubing. The shape and the inlet drop former determine the drop factor, which is the number of drops per millilitre of fluid.
Drop former
Inside or just above the drip chamber is a small calibrated nozzle that breaks the fluid into drops of a fixed volume. Macro-drip drop formers produce 10, 15 or 20 drops per millilitre; micro-drip drop formers produce 60 drops per millilitre. Drop factor is one of the most procurement-relevant attributes of an infusion set because it directly determines how the clinician calculates the flow rate.
IV tubing
Medical-grade PVC tubing, typically 1500–2000 mm long, 3.0 mm internal diameter for standard sets and 3.5–4.0 mm for blood transfusion sets. The tubing is the largest single source of leached plasticiser and is therefore the focus of DEHP-free upgrades. Length and diameter affect dead-volume, priming time, and the maximum gravity flow rate.
Roller clamp (and optional slide clamp)
A geared wheel on a frame that occludes the tubing by varying degrees, letting the clinician set flow rate visually against the drip chamber. A secondary slide clamp on the upstream side gives a binary on/off override that does not disturb the flow-rate setting. Roller clamps are still the dominant flow-control device on gravity sets even in pump environments, because they let the operator pre-prime the line.
Y-injection port (optional)
A latex or latex-free self-sealing rubber port in the side of the tubing that accepts a needle or needle-free connector for pushing additional medication into the running line. Luer Lock Y-port sets are the default for adult inpatient wards. Latex-free ports are mandatory in increasing numbers of hospital tenders.
In-line filter (optional)
A 0.2 micron or 1.2 micron particulate filter housed near the patient end of the tubing. Mainly used in paediatric, neonatal, total parenteral nutrition (TPN) and oncology infusions to remove particulate contamination. Adds cost and increases priming volume; only specify when the clinical use justifies it.
Luer connector
The tubing terminates in a male Luer connector — either Luer Slip (press-fit) or Luer Lock (threaded) — that mates with the IV cannula's female Luer hub or with an extension line. The choice has the same logic as on syringes; for a fuller treatment see Luer Lock vs Luer Slip Syringes: Which to Choose. For most adult IV use, Luer Lock infusion sets are the safer default because the joint is under sustained gravity load.
Needle or protective cap
Some sets ship with a hypodermic needle pre-attached for direct vein puncture (older protocol); modern sets usually terminate in a capped Luer Lock that connects to a separate IV cannula.
3. Drop factor in plain numbers
The drop factor (drops/ml) is a moulded property of the drop former and cannot be changed without swapping the set. The clinical implication: the same prescription "infuse 1 litre of normal saline over 8 hours" yields different drop counts on different sets.
| Set type | Drop factor | Drops/min for 125 ml/hr | Typical use |
|---|---|---|---|
| Macro-drip (US default) | 10 drops/ml | ~21 drops/min | Adult fluid replacement, large volumes |
| Macro-drip (European default) | 15 drops/ml | ~31 drops/min | Standard adult IV across most ISO 8536-4 markets |
| Macro-drip | 20 drops/ml | ~42 drops/min | Some Asian and Middle East markets, blood |
| Micro-drip | 60 drops/ml | ~125 drops/min | Paediatrics, neonates, slow medication |
Mixing macro and micro sets within the same ward is a known cause of medication errors, because clinicians who calculate by counting drops will over- or under-deliver if they grab the wrong set. Most hospital procurement standardises on one macro drop factor (typically 20 drops/ml in Asia, 15 drops/ml in EU, 10 drops/ml in US) for general use, and stocks a separate clearly-labelled micro-drip set for paediatric trolleys.
4. Set types and when to specify each
Standard Luer Slip infusion set
The volume-leader SKU. Macro-drip, no Y-port, terminating in a Luer Slip connector. Used in routine adult inpatient IV therapy in markets where Luer Slip is still the default and where the tubing run is short and stable. Lowest unit cost. Luer Slip infusion sets are typical of older Asian and Latin American hospital protocols.
Standard Luer Lock infusion set
Same anatomy with a threaded Luer connector. The mechanical lock prevents accidental disconnection between the set and the IV cannula, which matters because the joint sits at the patient's hand or arm and is under both gravity load and patient movement. Luer Lock infusion sets are the default in EU and most modern hospital tenders.
Y-port (Y-site) infusion set
Adds a self-sealing Y-injection port to the standard Luer Lock set, enabling secondary medication delivery, blood draw and piggyback infusion through the same line. Specify Luer Lock Y-port sets for adult ward, ICU, oncology and emergency department stock. The cost premium over a plain set is typically 8–15%.
Burette / big-chamber infusion set
Has a 100 ml or 150 ml graduated measuring chamber between the spike and the drip chamber. The clinician fills the chamber with the volume to be delivered, then closes the upper line — preventing accidental over-infusion if the patient is left unattended. Big-chamber burette sets are mainly stocked in paediatrics and neonatal ICU. They are governed by ISO 8536-10 in addition to 8536-4.
Blood transfusion set
Built around the same anatomy but with a 170–200 micron screen filter inside the drip chamber and wider tubing for higher flow at lower shear (to prevent haemolysis). Blood transfusion sets are governed by ISO 1135-4 and must be stocked as a separate SKU from IV infusion sets — a confused substitution can either filter out essential platelets (if a 0.2 micron in-line filter set is mistakenly used) or pass clots through to the patient (if a standard set without the screen is used for blood).
Pump-dedicated infusion set
Designed for a specific infusion-pump model. The tubing is engineered to fit the pump's peristaltic mechanism with the right occlusion characteristics; using a generic gravity set in a pump usually voids accuracy specifications. Pump sets are governed by ISO 8536-12. Always source pump sets from the pump manufacturer or from a vendor whose set is explicitly validated for your pump model.
5. Material choices: DEHP-free and latex-free
The two material attributes that drive most modern hospital tender language are plasticiser type and latex content.
DEHP-free
DEHP (di-2-ethylhexyl phthalate) is a phthalate plasticiser used to make medical PVC flexible. It can leach from tubing into infused fluid, especially with lipophilic medications (lipid emulsions, propofol, paclitaxel, cyclosporine), blood products, and during prolonged infusion. EU MDR (2017/745) classifies DEHP-containing devices as requiring specific labelling where they may be used with vulnerable populations (paediatrics, neonates, pregnant patients).
DEHP-free infusion sets use alternative plasticisers — typically TOTM (trioctyl trimellitate), DEHT, DINCH, or non-PVC tubing systems. The unit-cost premium is typically 10–25%; the technical performance is otherwise equivalent. For EU public hospital tenders, DEHP-free is increasingly written as a hard requirement; for most other markets it is a quality differentiator.
Latex-free
The Y-injection port is the main latex-containing component on a traditional infusion set. Latex-free Y-ports use a thermoplastic elastomer (TPE) or polyisoprene-free synthetic rubber. Hospital-acquired latex allergy was the primary clinical driver. Latex-free is now standard in most modern catalogues; if your supplier ships latex-containing ports, the carton must be clearly labelled "contains natural rubber latex" under FDA and EU rules. Specify latex-free explicitly in your purchase order to avoid ambiguity.
6. Compatibility with other devices
An infusion set is one component in a chain that runs from the IV bag to the patient's vein. The set must be compatible with the upstream and downstream devices in your protocol.
- IV bags and bottles. Match spike type (vented vs non-vented) to the container. Most flexible PVC bags use a non-vented spike; glass bottles and rigid plastic require vented.
- IV cannula / catheter. The Luer connector at the patient end must match the cannula hub. IV cannulas are universally Luer compatible (6% taper) but locking behaviour differs between Luer Slip and Luer Lock. Standardise the set–cannula connector type per ward.
- Infusion pump. A standard gravity set is not necessarily a valid pump set; check the pump manufacturer's compatibility list.
- Extension lines and three-way stopcocks. All must share the same Luer connector type, and the in-line dead volume must be calculated for accurate medication delivery in low-volume paediatric infusions.
- Filters and inline devices. A 0.2 micron in-line filter cannot be used with blood, lipid emulsions or particulate medications. Match the filter spec to the medication, not just the set type.
- Syringes for piggyback or push delivery. Y-port mates with any Luer connector, but for high-pressure injection (contrast, viscous medication) use a Luer Lock syringe to prevent disconnection at the port.
7. Procurement economics: MOQ, lead time, packaging
MOQ
From most Chinese manufacturers, MOQ for standard Luer Slip or Luer Lock infusion sets is typically 30,000–50,000 sets per SKU per production run. Y-port, burette and blood transfusion variants usually start at 20,000–30,000 sets because they are produced on slower assembly lines. DEHP-free or latex-free upgrades do not change MOQ but add to unit cost.
Lead time
25–35 days production after artwork sign-off and deposit. Sea freight from Qingdao or Shanghai adds 25–40 days depending on destination. Air freight cuts this to 7–10 days but at multiples of the freight cost; usually only justified for restocking after a stock-out.
Packaging
Standard packaging is individual blister film (PE or paper-Tyvek), with 25–50 sets per inner box and 200–400 sets per master carton. Outer carton labels carry the lot number, sterilisation method, expiry date, and ISO compliance reference. OEM-printed packaging is typically a one-time tooling fee per artwork (USD 200–500) rather than a per-unit premium.
Shelf life
36 months from manufacturing date is standard; some PVC-free or DEHP-free constructions reduce this to 24 months. Always insist on shelf life remaining at point of delivery, not at point of manufacture — there is a meaningful difference for slow-moving SKUs and for buyers who hold strategic stock.
8. Common buyer mistakes
- Specifying drop factor by region rather than by SKU. "European drop factor" means different things to different vendors. Always write the explicit number — 15 drops/ml or 20 drops/ml.
- Buying a generic infusion set for a specific pump. Pump sets are a separate category. Using gravity sets in a pump usually voids the pump's accuracy specifications.
- Mixing macro and micro within the same SKU line. Always distinct SKUs. Common cause of paediatric medication errors.
- Substituting blood transfusion sets for IV infusion sets. The 170 micron filter changes the flow profile and is required for blood; the 0.2 micron filter on a TPN set will trap blood cells and cause haemolysis.
- Accepting "DEHP-free packaging" as DEHP-free product. The plasticiser is in the tubing, not the wrapper. Insist on tubing material spec and a manufacturer letter confirming DEHP-free formulation.
- Latex Y-port without disclosure. Latex-free is the safe default; if your supplier ships latex without prominent labelling, both you and the hospital have a regulatory exposure.
9. Procurement checklist
Before issuing a PO for a new infusion-set SKU or a new vendor, confirm:
- Standard reference: ISO 8536-4 for IV sets, ISO 1135-4 for blood, plus any sub-part for burette / pump / paediatric variants.
- Drop factor written as an explicit number of drops/ml.
- Spike type (vented / non-vented or universal removable cap).
- Y-port presence and material (latex-free required for most modern tenders).
- Connector type (Luer Slip or Luer Lock) at the patient end.
- Tubing length (typically 150–200 cm) and internal diameter.
- Plasticiser system (DEHP-containing / DEHP-free) with manufacturer declaration.
- Latex content statement for every elastomeric component.
- Sterilisation method (EO, gamma) with sterility validation report.
- EO residual report meeting ISO 10993-7 limits.
- ISO 13485 quality system certificate, valid expiry; CE marking under EU MDR for European destinations; country-specific registration for ANVISA, COFEPRIS, SFDA, NAFDAC etc.
- Shelf life remaining at point of delivery (target 30+ months).
- OEM packaging artwork lead time and any plate-making fee.
For the broader supplier-vetting workflow that surrounds these technical points, see How to Choose a Disposable Syringe Manufacturer for Bulk Import — the same principles apply to infusion-set manufacturers.
10. Frequently asked questions
What are the main components of a standard IV infusion set?
A standard gravity-feed IV infusion set has eight functional components: a piercing spike that inserts into the IV bag or bottle, an air vent (on vented sets), a transparent drip chamber that lets the clinician count drops and trap air, a drop former that defines the drop size, an IV tubing line (typically 150–200 cm of medical-grade PVC), a roller clamp for flow control, an optional Y-injection port for adding medication mid-line, and a Luer connector at the patient end that mates with the IV cannula or extension line.
What is the drop factor of an infusion set and why does it matter?
Drop factor is the number of drops the set delivers per millilitre of fluid, defined by the geometry of the drop former. Macro-drip sets are typically 10, 15 or 20 drops/ml and are used for adult fluid replacement and rapid infusion. Micro-drip sets deliver 60 drops/ml and are used for paediatrics, slow medication infusion, and any application where dose accuracy at low flow rates matters. Confirm the drop factor on your spec sheet — different countries and manufacturers default to different values.
What is the difference between a vented and non-vented infusion set?
A vented infusion set has a small air vent on the spike that lets air enter the bottle as fluid drains, which is needed for rigid glass or hard plastic IV bottles. A non-vented set is used with collapsible plastic IV bags, where the bag itself collapses as fluid leaves, so no replacement air is required. Using a non-vented set on a rigid bottle will stop the flow as a vacuum builds; using a vented set on a soft bag is harmless but wasteful.
What is a Y-port and when should I specify one?
A Y-port (sometimes called a Y-site or Y-injection port) is a side-access port moulded into the IV tubing that allows clinicians to inject additional medication, draw blood, or piggyback a secondary infusion without disconnecting the primary line. Specify Y-port for any clinical area where multiple medications are administered through a single IV line — most adult inpatient wards, ICU, oncology and emergency department settings.
What does DEHP-free mean for an infusion set?
DEHP (di-2-ethylhexyl phthalate) is a plasticiser commonly used to make medical-grade PVC tubing flexible. Recent EU MDR regulation and many hospital tenders increasingly require DEHP-free tubing because DEHP can leach into the infused fluid, especially when the medication is lipophilic (lipid emulsions, certain anti-cancer drugs, blood products) and during prolonged infusion. DEHP-free sets use alternative plasticisers such as TOTM or DINCH that do not have the same leaching profile.
What is a burette or big-chamber infusion set used for?
A burette set has a large graduated measuring chamber (typically 100 ml or 150 ml) between the spike and the drip chamber, allowing the clinician to pre-measure a precise fluid volume before delivery. They are mainly used in paediatrics and neonatal care to prevent inadvertent over-infusion, and for accurate small-volume medication delivery. Burette sets are governed by the same ISO 8536 standard family as standard sets but are a separate SKU with its own MOQ and lead time.
Are blood transfusion sets the same as IV infusion sets?
No. A blood transfusion set has a built-in 170–200 micron filter in the drip chamber to capture clots and debris, has wider tubing to prevent haemolysis at typical flow rates, and is usually marked with the standard ISO 1135-4 rather than ISO 8536-4. A blood set is single-use only and should not be substituted for a standard IV infusion set, even though both terminate in a Luer connector. Always specify them as separate SKUs in tender documents.
What is the typical MOQ for IV infusion sets from a Chinese manufacturer?
Typical MOQ for standard Luer Slip or Luer Lock infusion sets is 30,000–50,000 sets per SKU. Y-port, blood transfusion and burette variants usually start at 20,000–30,000 sets because they are produced on slower assembly lines. DEHP-free or latex-free upgrades do not change MOQ significantly but add 10–25% to unit cost. OEM-printed packaging typically adds a one-time tooling fee but no MOQ premium.
11. Summary and how to request a quote
Infusion sets look interchangeable at a glance but break down into ten or more distinct procurement categories once you account for connector type, drop factor, side-port configuration, plasticiser system and ISO standard reference. Specifying a set by SKU number rather than by clinical use is the fastest way to make sure you get the same product reorder after reorder.
As a manufacturer of the full infusion-set family — Luer Lock, Luer Slip, Y-port, burette / big-chamber and blood transfusion sets — we ship to distributors and hospital networks in 50+ countries. Send us your specification (drop factor, connector type, Y-port, DEHP/latex requirements, packaging, destination market) and we will respond within one working day with applicable certifications, MOQ, lead time and a tiered quote. Request a quote
Related reading
- Luer Lock vs Luer Slip Syringes: Which to Choose for Your Application
- Auto-Disable vs Standard Syringes: WHO PQ Requirements for Vaccination Procurement
- How to Choose a Disposable Syringe Manufacturer for Bulk Import (2026 Guide)
- CE vs ISO 13485 vs FDA — A Medical Device Importer's Compliance Guide
- Browse all infusion-set product pages — full size and variant range
