Auto-Disable vs Standard Syringes: WHO PQ Requirements for Vaccination Procurement
Published May 6, 2026 · 14 min read · By HEZE YINUO MEDICAL
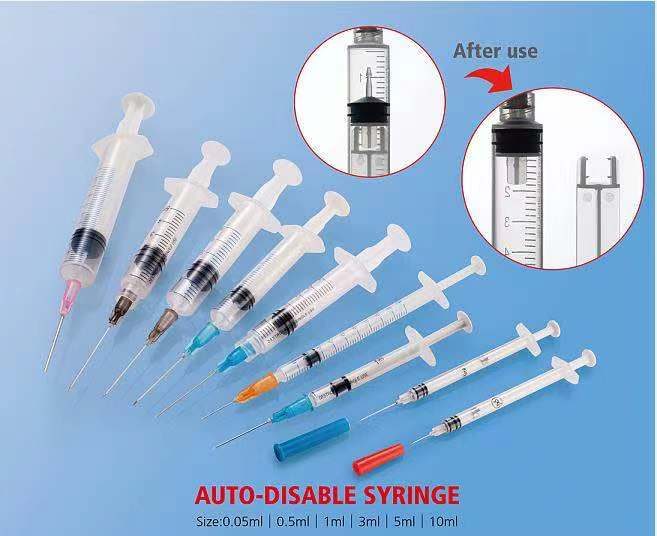
Quick answer. An auto-disable (AD) syringe is a single-use vaccination syringe with a mechanical lock that prevents the plunger from being pulled back after one delivery, so it cannot be refilled or reused. AD syringes are mandatory for WHO and UNICEF immunisation programmes and are specified in WHO PQS performance specification E08.IT.05 and the underlying ISO standard ISO 7886-3. They are not interchangeable with standard disposable syringes for therapeutic injection, and the procurement rules — prequalification listings, dose-volume SKUs, tender language, MOQ — are different. This guide walks through what changes once you start sourcing AD instead of standard syringes.
1. Why auto-disable exists: the unsafe-injection problem
For decades, syringe reuse in low-resource immunisation programmes was a major driver of bloodborne disease transmission. WHO's Safe Injection Global Network estimated that several billion injections per year carried a risk of HBV, HCV or HIV infection from reused syringes, with paediatric vaccination being one of the highest-volume risk environments. The mechanical solution was the auto-disable syringe: a syringe a healthcare worker physically cannot reuse, regardless of intent or training pressure.
Since 1999 WHO has recommended AD syringes as the preferred device for all immunisation, and since the early 2000s UNICEF Supply Division has procured AD syringes exclusively for the routine immunisation programmes it supports. Most low- and middle-income country EPI (Expanded Programme on Immunisation) tenders now specify AD as the default. As a buyer or distributor handling vaccination supply, this is not a clinical preference — it is a procurement requirement that has been written into tender language for two decades.
2. WHO PQS and the prequalification system
WHO operates a Performance, Quality and Safety (PQS) system for immunisation devices. PQS is a prequalification programme: WHO publishes a written specification for each device category, manufacturers submit products for testing against that specification, and listed products go into a public catalogue that procurement bodies (UNICEF, Gavi, country EPI) can buy from with confidence.
For auto-disable syringes the relevant document is PQS performance specification E08.IT.05, "Auto-disable syringe for fixed dose immunisation". It defines:
- The mechanical lock-out requirement (single-use, irreversible)
- Permitted dose volumes — most commonly 0.05 ml, 0.1 ml, 0.5 ml and 1.0 ml for childhood vaccines
- Connection type — Luer Slip with fixed needle (the needle cannot be detached, so the syringe cannot be combined with an alternative needle on a refilled barrel)
- Sterilisation by EO with ethylene oxide residual limits
- Packaging and labelling, including the WHO PQS reference number on the unit pack
- Performance testing referencing ISO 7886-3 "Sterile single-use syringes for fixed-dose immunisation"
A manufacturer who is not on the PQS list for E08.IT.05 cannot supply UNICEF, Gavi-funded country programmes, or most public-sector EPI tenders — even if their AD syringe is technically excellent and CE marked. PQS listing is a separate, additional certification on top of CE / ISO 13485, and it is the gating credential for vaccination contracts.
How to read a PQS catalogue entry
Each entry in the WHO PQS catalogue gives:
- PQS reference number (e.g. E08.IT.05/...)
- Product name and dose volume
- Manufacturer name and the manufacturing site that was audited (this matters — production at any other site is not covered by the listing)
- Date of prequalification and validity status
- Linked test reports (sterility, mechanical lock-out, biocompatibility, dose accuracy)
Always verify the PQS reference yourself on the WHO website before signing a procurement contract. Suppliers occasionally claim a PQS listing that has expired, has been suspended, or that belongs to a sister factory rather than the unit actually fulfilling your order.
3. AD vs RUP vs safety vs standard: a procurement-level comparison
The terms get conflated in informal procurement language. The distinctions matter because each device class is governed by a different ISO standard and serves a different clinical purpose.
| Class | Purpose | Standard | Reuse blocked? | Needlestick blocked? |
|---|---|---|---|---|
| Standard disposable | General-purpose injection | ISO 7886-1 | No | No |
| Auto-disable (AD) | Fixed-dose immunisation | ISO 7886-3 + WHO PQS E08.IT.05 | Yes (after first delivery) | No |
| Reuse-prevention (RUP) | Variable-dose general injection | ISO 7886-4 | Yes (after first delivery) | No |
| Safety / SIP | Sharps-injury prevention | ISO 23908; varies by region | Sometimes | Yes |
| Combined AD + safety | Immunisation in high-HIV settings | ISO 7886-3 + ISO 23908 | Yes | Yes |
Read the table from a tender-writing perspective: if your specification says "auto-disable", the supplier must deliver an ISO 7886-3 / E08.IT.05 product. If it says "safety syringe", any device meeting ISO 23908 needlestick-protection criteria qualifies, but it may not block reuse. The two requirements are independent — combine them only when your tender explicitly calls for both.
4. How AD syringes work mechanically
There are two main lock-out mechanisms in commercial AD syringes:
- Pre-set fixed-dose plunger. The plunger is locked at the dose volume from the moment of manufacture; the user pulls vaccine into the barrel, and after a single forward push to deliver, a clip or barb engages the barrel wall and prevents the plunger from being withdrawn again. This is the most common design and the cheapest to mould.
- Locking-after-injection plunger. The plunger remains free to move during dose drawing, but a one-way mechanism engages once full forward travel is reached. After delivery the plunger snaps or breaks at a designed weak point so that any attempt to draw back collapses or fractures the plunger rod.
Both designs are acceptable under WHO PQS E08.IT.05 as long as they pass the lock-out durability tests. The fixed-dose pre-set design is dominant in routine childhood immunisation (BCG, DTP, measles, polio) because it eliminates the dose-measurement variable and removes one source of healthcare-worker error.
Fixed-dose volumes you will see on tender specifications
- 0.05 ml — BCG (intradermal)
- 0.1 ml — special intradermal indications
- 0.5 ml — most paediatric vaccines (DTP, MMR, hepatitis B, pneumococcal, HPV)
- 1.0 ml — some adult formulations and specific country protocols
Each dose volume is a separate SKU in production, with separate moulds and separate PQS listings. A factory listed for 0.5 ml AD is not automatically listed for 0.1 ml — confirm the dose volumes on the supplier's PQS catalogue entry match exactly what your programme dispenses.
5. Procurement economics: what changes vs standard syringes
Unit cost
At factory gate, AD syringes typically cost 2x to 4x the unit price of an equivalent standard Luer Slip syringe. The premium reflects the moulded lock mechanism, tighter quality control (the lock-out feature must work 100% of the time, not 99.9%), and the cost of maintaining a WHO PQS-compliant quality system. In high-volume tender contracts the unit cost lands well under USD 0.10, which is why the price premium is accepted as the price of eliminating reuse risk in public health programmes.
MOQ and lot sizing
Typical MOQ from a Chinese manufacturer for a WHO PQS-listed AD syringe is 50,000 to 200,000 pieces per dose volume per production run. UNICEF and Gavi tender lots are normally measured in millions, so MOQ is rarely a constraint. For smaller country EPI programmes or NGO procurement, factories can usually run lots of 100,000 pieces; below that, expect a setup surcharge or wait for the factory to bundle your order with another small lot.
Lead time
Lead time is similar to standard syringes — 25–35 days production after deposit and artwork sign-off, plus 25–40 days sea freight. The PQS audit and listing process is on a different timescale (initial PQS prequalification can take 6–12 months for a new product), but that is upstream of any individual order: a factory either has the listing or it doesn't, and once listed, deliveries run on the same schedule as the rest of the catalogue.
Documentation
Every shipment of AD syringes for vaccination should arrive with:
- Certificate of analysis (COA) referencing the WHO PQS code
- Sterility validation report and EO residual report (ethylene oxide residue ≤ specified limits per ISO 10993-7)
- Batch-level dose-accuracy and lock-out test data
- ISO 13485 certificate, CE certificate, manufacturing site PQS letter
- Country-specific import documentation (Free Sales Certificate, Certificate of Origin, country MoH registration where applicable)
For the underlying compliance landscape — what each of these certificates means and how customs brokers interpret them — see our companion guide: CE vs ISO 13485 vs FDA: A Medical Device Importer's Compliance Guide.
6. Tender language: how AD specifications are written
A well-written AD syringe tender clause looks similar to this:
"Auto-disable syringe, single-use, fixed dose 0.5 ml, Luer Slip with fixed 23G × 25 mm needle, sterile (EO), latex-free. Performance compliant with ISO 7886-3:2020. WHO PQS-listed under specification E08.IT.05; supplier to provide current PQS reference number and copy of valid prequalification letter. Individual blister pack, 100 units per inner box, bulk carton labelled with batch number, manufacturing date, expiry date, and PQS reference. Shelf life minimum 36 months at point of delivery."
The procurement-critical phrases are "WHO PQS-listed under specification E08.IT.05" and "current PQS reference number". Without these, you are buying an AD-shaped syringe that may not actually be eligible for the funding programme paying for it, and your finance team can have the invoice rejected on audit.
Common buyer mistakes
- Specifying "auto-disable" without ISO 7886-3 or PQS reference. This produces quotes for a wide range of devices, some of which won't pass tender audit.
- Mixing dose volumes in one PO without specifying separate SKUs. Each dose volume needs its own line item, lot number and PQS reference; bundling them creates traceability problems on delivery.
- Asking for OEM-printed barrels at small lot sizes. Custom barrel artwork on AD syringes typically requires 1,000,000+ units due to specialised printing on PQS-locked moulds. For small lots, restrict customisation to outer packaging.
- Accepting CE-only without PQS for vaccination buyers. CE marking is necessary for EU markets but is not sufficient for UNICEF / Gavi / EPI procurement.
- Treating safety syringes as a substitute for AD. A safety syringe protects the clinician from needlestick but does not necessarily lock the plunger; for vaccination tenders this is a category error.
7. Combined AD + safety syringes
In high-prevalence HIV settings or where occupational sharps-injury data drives procurement (some Sub-Saharan African and Latin American programmes), tenders increasingly specify a combined device: AD lock-out plus retractable or shielded needle. These products meet ISO 7886-3 and ISO 23908 simultaneously and are roughly 3–5x the unit cost of a baseline AD syringe.
If your tender requires both, confirm that the manufacturer's PQS listing covers the combined device — not a separate AD listing and a separate safety listing. PQS treats combined-function products as their own category. Standalone safety syringes for therapeutic use are governed by ISO 23908 and do not need PQS for non-immunisation indications.
8. Where AD syringes don't fit
AD syringes are not a universal upgrade. They are wrong for:
- Variable-dose therapeutic injection. Pain management, antibiotic dosing, insulin titration — anything that needs a graduated barrel — requires a standard syringe or an RUP syringe (ISO 7886-4), not a fixed-dose AD.
- Aspiration / sample drawing. AD plungers are designed to lock against pull-back. Drawing blood or fluid samples requires the very motion AD prevents. Use a blood collection needle with a standard syringe or a vacuum tube system.
- Reconstitution and pharmacy compounding. Mixing multi-dose vials, drawing from ampoules, and transferring between containers all need free plunger movement. Use standard Luer Lock syringes for reconstitution and reserve AD for the actual delivery to the patient.
- Veterinary and laboratory use. No clinical reuse-prevention requirement, no PQS pathway, and the fixed-dose constraint is usually wasteful.
9. Procurement checklist for AD syringe tenders
Before issuing a purchase order or signing a tender award, confirm:
- Supplier holds a current WHO PQS listing under E08.IT.05 with valid expiry covering your dose volume and your delivery window.
- The listed manufacturing site is the site that will produce your order (some groups list one site and ship from another).
- Supplier holds ISO 13485:2016 certification with valid expiry.
- Supplier holds CE marking under EU MDR (required if delivering to EU, useful as a quality indicator elsewhere).
- Country-specific registrations are in place where required: ANVISA (Brazil), COFEPRIS (Mexico), SFDA (Saudi Arabia), NAFDAC (Nigeria), MFDS (Korea), etc.
- Sample testing pre-shipment: pull 100 units, deliver each through the lock-out cycle, attempt re-aspiration. Zero successful re-aspirations is the only acceptable result.
- Batch-level certificate of analysis and EO residual reports for every shipment.
- Shelf life of at least 36 months remaining at point of delivery (not at point of manufacture).
- Outer-carton labelling with PQS reference, batch number, expiry, and storage conditions.
For the underlying supplier-vetting workflow, see our broader guide: How to Choose a Disposable Syringe Manufacturer for Bulk Import.
10. Frequently asked questions
What is an auto-disable (AD) syringe?
An auto-disable syringe is a single-use disposable syringe with a built-in mechanism that prevents the plunger from being drawn back after the dose has been delivered, so the syringe cannot be reused. WHO and UNICEF require AD syringes for all childhood immunisation programmes to eliminate the risk of needle reuse and bloodborne pathogen transmission.
What is the difference between AD, RUP and safety syringes?
AD (auto-disable) syringes are designed for fixed-dose immunisation and lock the plunger after a single delivery; they are governed by ISO 7886-3. RUP (reuse-prevention) syringes are general-purpose syringes with a similar lock-out feature for variable doses, governed by ISO 7886-4. Safety syringes (sometimes called sharps-injury-prevention syringes) protect the clinician from needlestick injury via a retractable or shielded needle, but do not necessarily prevent reuse. Procurement specifications usually call for AD for EPI vaccination, safety for therapeutic injections in high-risk settings.
What is the WHO PQS standard E08.IT.05?
WHO PQS specification E08.IT.05 defines the performance requirements for auto-disable syringes used in routine and campaign immunisation. It mandates a single-use mechanism, fixed dose volumes (commonly 0.05 ml, 0.1 ml, 0.5 ml and 1.0 ml), Luer Slip with fixed needle, sterile EO, and documented testing under ISO 7886-3. Manufacturers seeking UNICEF or Gavi supply contracts must hold a WHO prequalification (PQS) listing for the specific product code.
Are AD syringes more expensive than standard syringes?
Yes. At factory gate, AD syringes typically cost 2-4 times the unit price of an equivalent standard disposable syringe, because of the moulded lock mechanism and tighter QA. However, for fixed-dose vaccination at scale the cost-per-dose is still well under USD 0.10 in bulk, and the public health benefit (eliminating reuse risk) is the reason the price premium is accepted by tender buyers.
What MOQ should I expect for AD syringes from a Chinese manufacturer?
Typical MOQ for WHO PQS-listed AD syringes is 50,000 to 200,000 pieces per dose volume per production run. For UNICEF or Gavi tender lots the order size is usually in the millions, so MOQ is rarely a constraint at that scale. For smaller country EPI orders or NGO procurement, factories can often run lots of 100,000 pieces; below that, expect a setup surcharge.
Can I use an AD syringe for therapeutic injections?
Technically yes, but it is wasteful. AD syringes are pre-set to a fixed dose volume, so a 0.5 ml AD syringe can only deliver 0.5 ml. For variable-dose therapeutic use, choose a reuse-prevention (RUP) syringe (ISO 7886-4) which has the lock-out feature but is graduated for any dose, or a standard syringe with a safety needle for needlestick protection.
How do I verify a manufacturer is on the WHO PQS list?
Search the WHO PQS catalogue by product code or manufacturer name on the WHO Performance, Quality and Safety website. Each listed product has a unique PQS reference, valid prequalification date, dose volume, and the manufacturing site that has been audited. Insist that the PQS reference number appears on the certificate of analysis your supplier provides for each shipment.
11. Summary and how to request a quote
Auto-disable syringes are not a more expensive version of standard syringes — they are a different procurement category, governed by a different ISO standard, listed in a different catalogue, and bought through a different tender process. The decision tree is straightforward: if you are supplying childhood or campaign immunisation under any WHO, UNICEF, Gavi or EPI funding line, you need AD with a current PQS listing on the right dose volume. For everything else — therapeutic injection, reconstitution, sampling, dental, veterinary, laboratory — a standard Luer Lock, Luer Slip or safety syringe is the correct device.
As a manufacturer of auto-disable syringes, 1ml vaccine syringes, safety syringes and the full standard Luer Lock / Luer Slip range, we ship to government procurement programmes and NGO supply chains in 50+ countries. Send us your dose volume, target quantity, destination market and any tender language you need to satisfy, and we will respond within one working day with applicable certifications, MOQ, lead time and a tiered quote. Request a quote
Related reading
- Luer Lock vs Luer Slip Syringes: Which to Choose for Your Application
- How to Choose a Disposable Syringe Manufacturer for Bulk Import (2026 Guide)
- CE vs ISO 13485 vs FDA — A Medical Device Importer's Compliance Guide
- Auto-Disable Syringes — full size range, specifications and quote form
- 1ml Vaccine Syringes — Luer Slip and Luer Lock variants
